
GMP Cleanroom Consultancy Services UK
W2 Cleanroom Consulting provides structured GMP cleanroom consultancy services across the United Kingdom. We support pharmaceutical manufacturers and NHS aseptic services with Annex 1 aligned facility design review, contamination control strategy integration and full validation lifecycle planning.
Facility Design & Layout
We provide risk-based cleanroom layout and sterile facility design review aligned to GMP and Annex 1 expectations. Our assessments focus on material and personnel flows, zoning strategy, pressure differentials, HVAC considerations and contamination control integration.
Includes:
Concept and detailed layout planning
Airflow and pressure cascade mapping
Material and personnel flow analysis
Room data sheets and zoning plans


Validation & Qualification
We support cleanroom validation and qualification aligned to Annex 15 lifecycle principles. Our approach defines clear DQ, IQ, OQ and PQ strategy, commissioning boundaries and inspection-facing documentation structures.
Includes:
Validation Master Plans (VMPs)
Protocols and acceptance criteria
IQ/OQ/PQ testing and reporting
Traceability and summary reporting
We support GMP quality system strengthening within sterile manufacturing environments, including document control frameworks, deviation and CAPA processes, SOP alignment and inspection readiness preparation.
Includes:
QMS setup and gap analysis
SOP suite development and document control
CAPA and deviation management
MHRA inspection readiness reviews
Quality Governance & GMP Support




Service Categories
Equipment & URS Development
We develop regulatory-ready User Requirement Specifications (URS) for cleanroom systems, isolators, HVAC, barrier technology and automation platforms. Each URS is structured to support Design Qualification and downstream verification.
Includes:
URS authoring and approval process
Equipment tender and vendor alignment
FAT/SAT scope definition
Data integrity and electronic batch record integration
Cleanability and material compatibility requirements
Environmental monitoring considerations
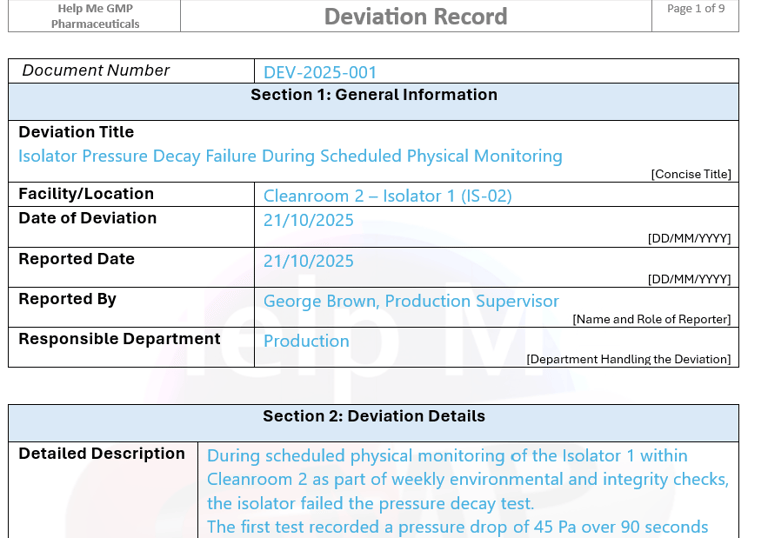
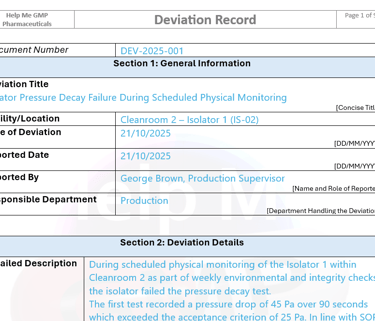
Cleanroom projects vary in complexity, regulatory risk and governance structure. Our consultancy model is adapted to the specific sterile process, facility classification and organisational maturity of each client.

FAQ
What is cleanroom?
A cleanroom is a highly controlled environment where temperature, humidity, and airborne particles are tightly regulated to protect products and processes. Cleanrooms are essential in aseptic manufacturing, pharmaceutical production, and other sectors that demand contamination control.
Why is validation important?
Validation ensures your cleanroom, equipment, and processes perform consistently and meet regulatory requirements such as EU GMP Annex 1. It’s the formal evidence that your systems work as intended — safeguarding both product quality and patient safety.
How do you optimize workflows?
We analyse material and personnel flows, equipment placement, and process steps to remove bottlenecks and reduce contamination risk. Our consultants bring real-world experience from operating within cleanrooms, ensuring every recommendation is practical, compliant, and achievable.
What services do you offer?
We provide a full range of cleanroom consulting services, including:
Facility design and layout planning
Validation strategy and documentation (VMP, IQ/OQ/PQ)
Quality governance and GMP support
Section 10 aseptic consultancy for NHS services
URS development for isolators, HVAC, and equipment
Who are your clients?
We work with NHS aseptic units, pharmaceutical manufacturers, and healthcare organisations across the UK. Our consultants have delivered cleanroom projects from early design through to validation and inspection readiness.
How can I contact you?
You can reach us via info@w2cleanrooms.com or through our contact form on the website. We’re happy to discuss upcoming projects, validation support, or general GMP consultancy needs.
Contact
Get in touch
info@w2cleanrooms.com
© 2025. All rights reserved.
Our Brands
Free GMP Training
Click for more info





